The Effect of a New Dietary Mineral Product on Body Composition and Weight in Overweight and Obese People. The Results from a Comparative Randomized 30-Days Study
Hesselberg JO, Wahl A and Olsen TE
DOI10.21767/2471-8203.100015
Hesselberg JO*, Wahl A and Olsen TE
HW Research and Development, Sogstikroken 14, Drøbak, 1446, Norway
- *Corresponding Author:
- Hesselberg JO
HW Research and Development
Sogstikroken 14, Drøbak
1446, Norway
Tel: 004790071673
E-mail: janole@hw-research.no
Received date: May 05, 2016; Accepted date: May 30, 2016; Published date: June 04, 2016
Citation: Hesselberg JO, Wahl A, Olsen TE (2016) The Effect of a New Dietary Mineral Product on Body Composition and Weight in Overweight and Obese People. The Results from a Comparative Randomized 30-Days Study. J Obes Eat Disord 2:3. doi: 10.21767/2471-8203.100015
Copyright: © 2016, Hesselberg JO, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The efficacy of a new dietary mineral product based on natural ingredients was investigated in this comparative randomized control group study. The formulation is based on the middle calcareous layer of oyster shells, in homeopathic and traditional medicine known as calcarea carbonica, as well as iodine, magnesium and chromium. Fourteen none-smoking overweight subjects (BMI ≥ 25.5 kg/m2) were invited to participate in the study. The results show that during a treatment period of 30 days significant reduction are seen both in body weight (BW) and body mass index (BMI). There was also a positive change in waist- and hip circumference, although this change was not statistically significant. Previous studies have shown that all four main ingredients in the formulation might have positive effects on body weight and body composition. This product shows promising results, but additional research is warranted to further clarify the mechanisms responsible for this effect.
Keywords
Dietary products; Natural ingredients; Homeopathy; Weight loss; Calcium; Iodine; Magnesium; Chromium
Introduction
Obesity is a major problem in many countries, and a risk factor for a number of diseases, including diabetes, cardiovascular diseases and cancer, according to the World Health Organization. Worldwide obesity has more than doubled since 1980, and most of the world's population now live in countries where overweight and obesity kills more people than underweight [1]. Changes in food intake and physical exercise are the only effective measures in the long run, but to help initiating weight reduction there are a number of agents, drugs and natural substances available. Studies have shown that certain minerals can have a positive effect on weight reduction.
Calcium has been identified as playing an important role in the regulation of energy and lipid metabolism [2]. Observational studies have demonstrated that calcium intake is inversely associated with body weight [3-5] and type 2 diabetes [6]. However, results from randomized controlled trials to evaluate the effect of supplemented calcium on weight reduction and metabolic profiles remain controversial [7,8].
Iodine is an essential component of thyroid hormones that are required for normal metabolic processes. The major sources of dietary iodine in most western countries are dairy products, grains, and iodized salt [9]. Studies have shown that individuals following a restricted diet could be at risk of inadequate dietary iodine intake [10].
Magnesium is a cofactor in a number of key enzymatic reactions in the body [11]. It plays a crucial role in glucose metabolism and insulin homeostasis [12,13]. Deficient magnesium intake may be a risk factor for the development of type 2 diabetes [14-16] and metabolic syndrome [17,18].
Chromium is an essential nutrient involved in the regulation of carbohydrate and lipid metabolism [19]. A recent metaanalysis concludes that evidence from available RCTs shows that chromium supplementation generates statistically significant reductions in body weight [20].
To our knowledge it has never been conducted a clinical trial aimed to determine the effect of these four compounds combined, on weight loss.
The aim of this study was to determine the efficiency of a new natural weight-reduction food supplement containing all these minerals, with the trade name Restart® (NorMed Pharma AS, Oslo, Norway).
Methods
Subjects
Fourteen overweight non-smoking individuals of both sexes were recruited for the study through an advertisement on a website. The volunteers had a body mass index(BMI) in the 26.0–39.6 range, but where otherwise healthy. All participants provided written informed consent before entering the study, after having received information about the study procedures. The study was conducted according to the principles of the revised Declaration of Helsinki, Good Clinical Practice and local regulation.
Study design
The study was carried out as a randomized control group study. Nine subjects were treated with the active preparation and five were in the control group. Unfortunately four subjects dropped out of the study (three from the control group, one from the intervention group). They did not attend for the follow-up after 30 days, and when we contacted them, two said they lacked the motivation to come and two were unable. These subjects are not included in the analysis. The treatment period was 30 days. Participants should not use any drugs for chronic diseases or be on any weight reduction treatment during the study period. No diet advice or recommendations with respect to exercise were given to the participants prior to inclusion in the study.
Demographic data
Anthropometric measurements, including height, weight, and waist- and hip circumference were made initially, and at the follow-up after 30 days. The body mass was measured to an accuracy of 0.1 kg and height was measured to the nearest cm. Hip and waist circumference was measured in a standardized way on exactly the same position on each of the visits, but not by the same person.
Table 1 presents the baseline anthropometric parameters for the two groups of participants, which reveal that both groups were comparable at the start of the study with respect to the parameters of interest.
| Control group (n=2) | Intervention group (n=8) | |
|---|---|---|
| Age | 40.5 | 34.7 |
| Female (%) | 2 (40%) | 8 (89%) |
| Waist circ. | 106.0 | 110.9 |
| Hip circ. | 112.5 | 116.4 |
| Weight (kg) | 85.4 | 93.9 |
| Height (cm) | 160.0 | 168.1 |
| BMI (kg/m2) | 33.4 | 32.9 |
Table 1: Baseline mean anthropometric parameters for intervention and control group.
Treatment
The investigational preparation used in this study was Restart® from Nor-Med Pharma, Oslo, Norway. The product is defined as a food supplement, and each tablet contains the following active ingredients: Calcium hydrogen carbonate (Calcarea carbonica), calcium silicate, potassium iodide, chromium (III) chloride and magnesium. According to the manufacturer the daily dosage (2 tablets) contains: Calcium 450 mcg (55% *) Iodine 150 mcg (100% *) Magnesium 150 mcg (50% *) Chromium 50 mcg (125% *) * % of ref. value. The supplement was not on the market when the study took place. The dosage was two tablets in the evening swallowed with water. No formal dose-response studies have been carried out.
Statistical analysis
Significant differences between the intervention group and the control group were measured by using two-factor repeated measures analysis of variances (ANOVA). A P-value < 0.05 was considered to be statistically significant.
Results
A total of 10 subjects concluded the study, eight in the intervention group and two in the control group. Four subjects dropped out of the study, one in the intervention group and three in the control group, due to personal reasons.
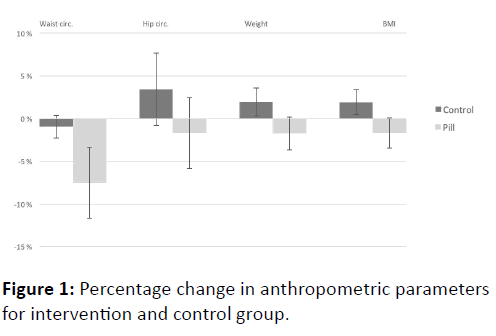
Table 2 and Figure 1 shows the changes in weight, BMI and waist- and hip-circumference in the two groups during the study. The weight loss and BMI reduction was statistically significant in the intervention group (p<0.05). Hip and waist circumference also showed a positive change, although this was not statistically significant. None of the participants reported any side effects that could be related to the treatment they received.
| Groups | Control group (n=2) | Intervention group (n=8) | p-value |
|---|---|---|---|
| Waist circ. | -1 (1.4) | -7.8 (4.3) | ns |
| Hip circ. | 4 (4.9) | -1.9 (4.8) | ns |
| Weight (kg) | 1.7 (1.4) | -1.6 (1.7) | .03 |
| BMI (kg/m2) | .6 (.5) | -.6 (.6) | .03 |
Table 2: ANOVA analysis of post intervention change for intervention and control group.
Discussion
In this study, participants treated with a combination of calcium, iodine, magnesium in dosages of respectively 450, 150, 150 and 50 mcg, had a statistically significant reduction in body weight and body mass index compared to a control group. The same statistically significant effect was not seen in either hip or waist circumference.
Strengths and limitations
This is likely the first clinical trial aimed to determine the effect of combined calcium, iodine, magnesium and chromium, on weight loss. A major limitation in this study, however, is the small number of subjects. The drop out after the initial registration resulted in a control group of only two participants. In addition the study was carried out as a simple randomized control-group study, so psychological effects should not be ruled out.
Possible mechanisms
Minerals like calcium, iodine, magnesium and chromium are vital for the regulation of energy and lipid metabolism, the regulation of thyroid hormones, glucose metabolism and insulin homeostasis. However, as this dietary supplement is a combination of four different minerals, it is difficult to determine the exact mechanisms behind the measured effect and the study design and methods do not allow for such inferences.
Conclusion
Obesity and overweight are serious health problems in most industrialized countries. A variety of approaches to combat this have been attempted, but despite this average body weight is still increasing. Despite obvious limitations that make it hard to draw clear conclusions from this study, the results add to promising results of previous studies on the effects of calcium, iodine, magnesium and chromium as weight loss supplements. Additional research on the effect of these four compounds combined is warranted to further clarify the mechanisms responsible for the observed effects.
Financial Support
This project has been financially supported by Nor-Med Pharma, Oslo, Norway.
References
- World Health Organization: Obesity and overweight, fact sheet No311, Geneva, Switzerland 2015.
- Jacqmain M, Doucet E, Despres JP, Bouchard C, Tremblay A (2003)Calcium intake, body composition, and lipoprotein-lipid concentrations in adults.Am J ClinNutr77:1448–1552.
- Bueno MB, Cesar CL, Martini LA, Fisberg RM (2008)Dietary calcium intake and overweight: an epidemiologic view.Nutrition 24:1110–1115.
- Elwood PC, Strain JJ, Robson PJ, Fehily AM, Hughes J, et al. (2005) Milk consumption, stroke, and heart attack risk: evidence from the Caerphilly cohort of older men.J Epidemiol Community Health59:502–505.
- Gonzalez AJ, White E, Kristal A, Littman AJ (2006)Calcium intake and 10-year weight change in middle-aged adults.J Am Diet Assoc 106:1066–1073.
- Pittas AG, Lau J, Hu FB, Dawson-Hughes B (2007)The role of vitamin D and calcium in type 2 diabetes. A systematic review and meta-analysis.J ClinEndocrinolMetab 92: 2017–2029.
- Major GC, Chaput JP, Ledoux M, St-Pierre S, Anderson GH, et al. (2008) Recent developments in calcium-related obesity research.Obes Rev9:428–445.
- Muscogiuri G, Sorice GP, Ajjan R, Mezza T, Pilz S, et al. (2012)Can vitamin D deficiency cause diabetes and cardiovascular diseases? Present evidence and future perspectives.NutrMetabCardiovasc Dis22:81–87.
- Pearce EN, Pino S, He X, Bazrafshan HR, Lee SL, et al. (2004) Sources of dietary iodine: bread, cows’ milk, and infant formula in the Boston area.J ClinEndocrinolMetab89:3421–3424.
- Pennington JA, Schoen SA (1996) Contributions of food groups to estimated intakes of nutritional elements: Results from the FDA total diet studies, 1982–1991.Int J VitamNutr Res 66:342–349.
- Wester PO (1987) Magnesium.Am J ClinNutr45:1305–1312.
- Paolisso G, Barbagallo M (1997) Hypertension, diabetes mellitus, and insulin resistance: the role of intracellular magnesium.Am J Hypertens10:346–355.
- Barbagallo M, Dominguez LJ, Galioto A, Ferlisi A, Cani C, et al. (2003) Role of magnesium in insulin action, diabetes and cardio-metabolic syndrome X.Mol Aspects Med24:39–52.
- Kao WH, Folsom AR, Nieto FJ, Mo JP, Watson RL, et al. (1999) Serum and dietary magnesium and the risk for type 2 diabetes mellitus: the Atherosclerosis Risk in Communities Study.Arch Intern Med159:2151–2159.
- Lopez-Ridaura R, Willett WC, Rimm EB, Liu S, Stampfer MJ, et al. (2004) Magnesium intake and risk of type 2 diabetes in men and women.Diabetes Care27:134–140.
- Song Y, Manson JE, Buring JE, Liu S (2004) Dietary magnesium intake in relation to plasma insulin levels and risk of type 2 diabetes in women.Diabetes Care27:59–65.
- Guerrero-Romero F, Rodriguez-Moran M (2002) Low serum magnesium levels and metabolic syndrome.ActaDiabetol39:209–213.
- Song Y, Ridker PM, Manson JE, Cook NR, Buring JE, et al. (2005) Magnesium intake, C-reactive protein, and the prevalence of metabolic syndrome in middle-aged and older U.S. women.Diabetes Care28:1438–1444.
- Albarracin CA, Fuqua BC, Evans JL, Goldfine ID (2008) Chromium picolinate and biotin combination improves glucose metabolism in treated, uncontrolled overweight to obese patients with type 2 diabetes. Diabetes Metab Res Rev 24: 41–51.
- Onakpoya P, Posadzki, Ernst E (2013) Chromium supplementation in overweight and obesity: a systematic review and meta-analysis of randomized clinical trials. Obes Rev 14: 496-507.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences